Hickman triple lumen catheter12/30/2023  There is a need to evaluate the value of these devices to the UK NHS by looking at clinical and cost-effectiveness. Therefore, the magnitude of this reduced risk is still unclear. The evidence is weak and the studies are heterogeneous, in terms of patient populations, methodological approach and definition of outcomes. However, there is some evidence that TIVAS may have the lower complication rate and lead to greater patient satisfaction with less interruption to treatment regimens ( Kulkarni et al, 2014). 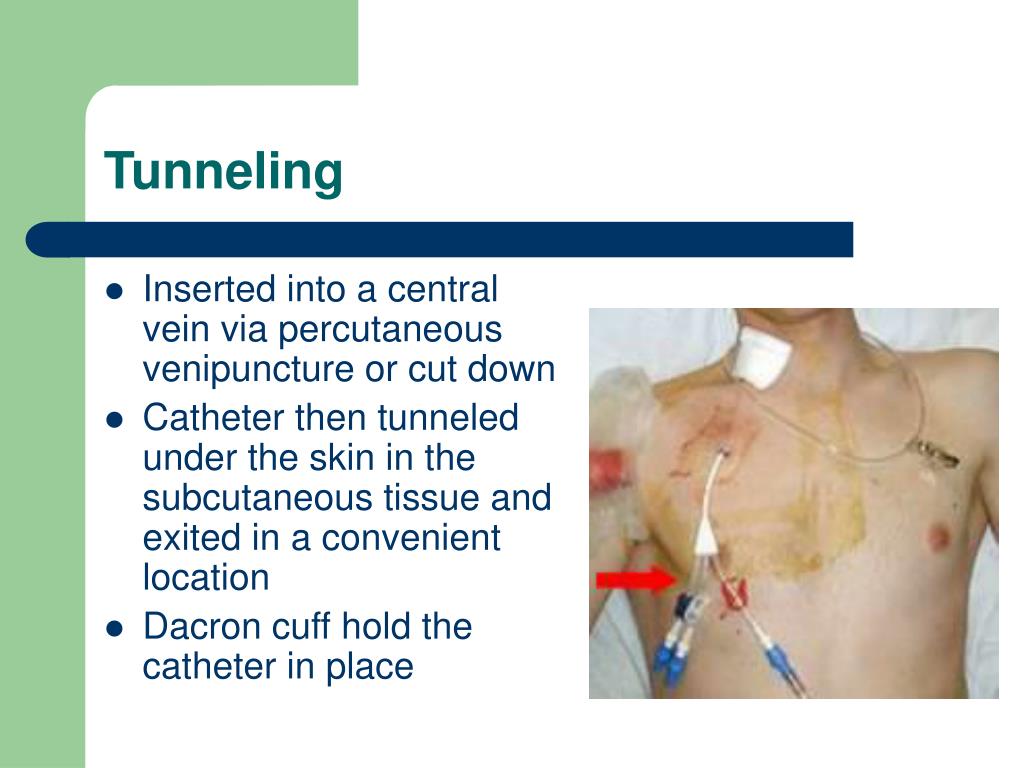
The TIVASs are more expensive, more complex and invasive to insert, and many healthcare staffs are unfamiliar with their aftercare. A recent informal survey (personal communications) of nine large UK cancer units indicated Hickman (58%) to be the most common followed by PICC (33%), with TIVAS only used in 9%. 
There are three main types of central device: (i) tunnelled central catheter commonly referred to as a Hickman (ii) peripherally inserted central catheter (PICC) and (iii) totally implanted venous access system (TIVAS) commonly referred to as a port ( Bishop et al, 2007). These advantages make central devices the obvious choice for longer drug regimes. When the catheter tip lies centrally in a large vein, the damage is mitigated due to rapid blood flow and large vessel diameter. This results in rapid exhaustion of the forearm veins, interruption to treatment, patient discomfort and a genuine fear of cannulation ( Cheung et al, 2009). Peripheral administration of chemotherapy frequently causes local vein irritation and thrombosis. When intravenous chemotherapy is needed it can either be given through a peripheral cannula (typically in a forearm vein) or through a central venous access device where the catheter tip is placed in a large central vein (typically the superior vena cava). A larger multicentre trial is needed to confirm these preliminary findings. Conclusions:Ĭompared with Hickman catheters, TIVAS may be the cost-effective option. However, there is much uncertainty associated with the results. The economic evaluation showed that Hickman arm was associated with greater mean cost per patient £1803 (95% CI 462, 3215), but similar quality-adjusted life years −0.01 (95% CI −0.15, 0.15) than the TIVAS arm. Quality of life based on the device-specific questionnaire was greater in the TIVAS arm for 13 of the 16 questions. In the Hickman arm, 28% of the devices were removed prematurely due to a complication compared with 4% in the TIVAS arm. Overall, 54% of patients in the Hickman arm suffered one or more complications compared with 38% in the TIVAS arm (one-sided P=0.068). Results:Ī total of 100 patients were randomised in a 3 : 1 ratio to receive a Hickman or a TIVAS. An economic evaluation was also carried out. Secondary end points included individual complication rates, time to first complication and quality of life. The primary end point was overall complication rate. This was a phase II prospective, randomised, open trial from two UK oncology centres. This pilot trial aimed to generate relevant data to inform the design of a larger definitive randomised controlled trial. However, it is unclear whether the higher costs may be offset by perceived greater health benefits. 
Compared with Hickman catheters, these devices are more expensive and complex to insert. In the United Kingdom, totally implantable venous access systems (TIVAS) are not routinely used. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
